Nematode Management Guides – Additional Resources
Root Lesion Nematode Behavior
Nematodes are animals so they have a nervous system and the ability to respond to a wide range of signals. Lesion Nematodes can’t see or hear, but their ability to sense chemical gradients in the soil is incredible. They are so small that their movement through soil is restricted to pore spaces which means the path to plant roots winds around soil particles packed together in the soil matrix. The size and number of pore spaces, and the hence the number of twists in the path, is determined by soil texture. Sandy soils provide the most direct route for nematodes and lose excess water which helps nematodes move as they cannot swim.

Credit: Carrie Lapolla in Williams, C. 2021. Soil Engineering: The relationship between soil texture and function https://www.sciencefriday.com/educational-resources/soil-texture/
Roots exude a bevy of chemicals that beacon nematodes. Some are common to all plants and some are very specific. The impact of this signal “call” from corn to RLN is illustrated below. Nematodes sheltered in a dead root sensed food was nearby when the dead root was placed in a Petri dish of healthy corn roots. RLN are too small to see, but the developing lesion two weeks later reveals that nematodes found a new home in the healthy root. The middle photo shows a root culture that has been completely invaded by RLN 20 weeks after being placed in the dish. RLN often leave roots that are fully colonized and may show the interesting behavior of forming clusters as in the photo on the right. Each cluster is made up of hundreds to several thousand RLN.

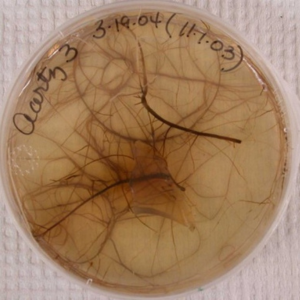
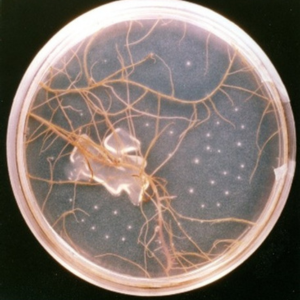
Photo credit: Ann MacGuidwin
State surveys for Root Lesion (RLN) and other nematodes
Survey data has been published for nematodes associated with soybean or corn rotated with soybean in 14 states. RLN was the first or second most prevalent nematode detected in 12 of the 14 surveys. Spiral and Stunt nematodes were very common, but they are not considered to be yield-damaging pests of soybean.
The surveys are summarized below with the crop sampled, years of the survey, and five most prevalent nematodes shown for each state.
Alabama: Crop = Soybean. 2008-2010. Top nematodes: Reniform 31%, RLN 16%, Root-Knot 13%, SCN 11%. (Sikora el al., 2011, Plant Health Progress DOI.10.1094/PHP-2011-1227-01-RS)
Arkansas: Crop = soybean. 1978-1986. Top five nematodes: SCN 67%, RLN 45%, Stunt 43%, Dagger 25%, Spiral 23% . (Robbins, et. al., 1987, Annals of Applied Nematology 1:55-55)
Delaware: Crop = soybean. 2019-2021. Top five nematodes: RLN 57%, Lance 56%, Spiral 54%, SCN 53%, Stunt 45%. (Kessler & Koehler, 2023. DOI.10.1094/PHP-07-22-0064-S)
Georgia Crop = soybean. 2021. Top five nematodes: Spiral 85%; RLN 71%, Stubby-Root 69%, Ring 54%, Root-Knot 49%. (Mitchem et al., 2023, Plant Health Progress DOI.10.1094/PHP-09-22-0096-S)
Illinois Crop = corn. 2018 & 2020. Top five nematodes: Spiral 98%, RLN 86%, SCN 67%, Stunt 33%, Dagger 13%. (Han et al., 2021, Plant Health Progress DOI.1094/PHP-01-21-0012-S)
Iowa Crop = corn. 2000-2010. Top five nematodes: Spiral 77%, RLN 51%, Dagger 42%, Lance 23%, Needle 8%. (Tylka et al., 2011, Plant Health Progress DOI.10.1094/PHP-2011-1205-01-RS)
Kansas Crop = wheat. 2007-2010. Top five nematodes: Stunt 81%, RLN 77%, Pin 28%, Spiral 8%, Dagger 4%. (Todd et al., 2014, Plant Health Progress DOI:10.1094/PHP-RS-13-0125)
Maryland: Crop = soybean. 2019-2021. Top five nematodes: RLN 57%, Lance 56%, Spiral 54%, SCN 53%, Stunt 45%. (Kessler & Koehler, 2023. DOI.10.1094/PHP-07-22-0064-S)
Michigan Crop = corn. 2018. Top five nematodes: Spiral 91%, RLN 81%, Stunt 79%, SCN 57%, Pin 21%. (Thapa et al., 2023, Journal of Nematology DOI.102478/jofnem-2023-0015)
Minnesota Crop = soybean. 1956-1957. Top five nematodes: Dagger 78%, Spiral 52%, Stunt 52%, RLN 41%, Pin 39%. (Taylor et al., 1958, Plant Disease Reporter 42:195-198)
N. Carolina Crop = soybean. 1994-1996. Top five nematodes: Spiral 100%, RLN 72%, SCN 71%, Stunt 62%, Stubby-Root 56%. (Koenning & Barker, 1998, Journal of Nematology 30(4s):569-576
N. Dakota Crop = corn. 2015-2016. Top five nematodes: Spiral 52%, Stunt 37%, Pin 31%, RLN 20%, SCN juveniles 9%. (Chowdhury et al., 2020, Canadian Journal of Plant Pathology DOI.10.1080/07060661.2019.1674384)
Pennsylvania Crop = soybean. 2018-2023. Top nematodes: RLN 80%, SCN 3%, Root-knot 2%. (Pennsylvania Soybean Board Research Report, 2023)
S. Carolina Crop = soybean. 1987 & 1989. Top five nematodes: Spiral 76%, RLN 67%, Stubby-Root 64%, Ring 45%, Root-Knot 28%. (Lewis et al., 1993, Journal of Nematology 25(4S):89-=894)
Wisconsin Crop = soybean & crops rotated with soybean. 2012-2021. Top five nematodes: RLN 84%, Spiral 70%, Dagger 33%, SCN 26%, Stunt 18%. (MacGuidwin et al., 2023, Journal of Nematology DOI.10.2478/jofnem-2023-0053)
Other Nematode Pathogens
Soil testing for RLN also recovers other nematode pathogens. Identification to the species level is so laborious that nematode diagnostic labs lump many species together into a common name. Common names for nematodes refer to their primary host, the plant symptom they cause, geography, or a defining shape or structure. Some species have their own common name such as the reniform nematode (Rotylenchulus reniformis), southern root knot nematode (Meloidogyne incognita) and the soybean cyst nematode (Heterodera glycines), but most often a common name refers to all of the species in a genus or taxonomic family, such as root lesion nematode.
- Nematodes named for the plant symptom they cause: Root Knot, Root Lesion, Stem, Stubby Root, Stunt, Sting
- Nematodes named for body shape: Cyst, Reniform, Spiral
- Nematodes named for an external body structure: Ring, Sheath
- Nematodes named for their straw-like sucking mouthparts (stylet): Awl, Dagger, Lance, Needle, Pin
SCN, Root Knot, Reniform, and Root Lesion (some species) often reach population densities sufficient to damage soybean. Lance, Dagger, and Sting nematodes occasionally reach population densities sufficient to damage soybean, particularly in Southern states. Awl, Pin, Ring, Sheath, Spiral, Stubby Root, and Stunt nematodes rarely reach population densities sufficient to damage soybean.
Lance (left), Dagger (middle), and Ring (right) nematodes. Photo credits: Lance and Ring; Jonathan Eisenback, Dagger; Penn State Department of Plant Pathology & Environmental Microbiology


Nematode Test Reports
Nematode tests report the number of nematodes detected per volume of soil (commonly 100 cubic centimeters) or per weight of root (grams). Samples submitted in the spring and fall are used to predict the impact of nematodes on yield for the current or next crop, respectively, based on damage threshold values. Some labs use damage thresholds that are generalized across crops, soil types, and even time of year. Others use thresholds that are specific to a particular set of conditions. In general, damage thresholds are lowest for preplant samples and highest for fall samples because winter-kill of nematodes must be taken into account to predict damage for next year’s crop. RLN damage thresholds for soybean tend to be lower than for corn, and higher than for vegetables.
When looking at numbers of RLN it is important to note that RLN produce fewer offspring than SCN, Root Knot, and Reniform nematodes. One study estimated the average female Pratylenchus penetrans produce 28 eggs over her lifetime, so population densities exceeding 1000 RLN per 100 cc of soil are exceptionally high and extremely rare. As a very general rule of thumb; 0 – 99, 100-200, and > 200 RLN per 100 cc soil are commonly considered to represent low, moderate, and high population densities, respectively, for samples collected after September 1.
Numbers of nematodes recovered per gram of root decreases as soybean plants develop up to the R4 or R5 growth stage. RLN reproduce continuously, but it is only after soybean root growth slows that the density of nematodes in roots increases. Diagnostic root samples collected at this point from symptomatic areas in the field suggest RLN is involved when population densities exceed 1000 nematodes per gram of root.
How do testing labs determine the number of Root Lesion Nematode (RLN) in a sample?
There are two basic procedures to remove nematodes from soil, with many minor variations.
The first procedure physically separates nematodes from soil by adding water and then filtering the soil-water mixture through sieves with different mesh sizes. Fine soil particles and organic matter are also collected so a sugar solution may be used to isolate nematodes as they float away from heavier material. All nematodes, including those that have died and eggs, are recovered by soil filtering. Some laboratories use automated devices that add water, mix, and filter soil from multiple samples simultaneously; other labs process one sample at a time by hand.
The second procedure relies on nematode movement, so only living and motile nematodes are recovered. Soil is placed in contact with a water reservoir and nematodes move downward, eventually falling into the water where they are collected. Many labs use a 48-hour incubation time, but shorter or longer times are not uncommon.
Nematodes are recovered from roots using an incubation method similar to that described for soil. Roots are placed in contact with a water reservoir and as nematodes exit, they fall into the reservoir and sink to the bottom. Some labs use the root fragments collected on a coarse-mesh sieve used in the filtering procedure while others cut roots from plants submitted with the soil sample.
All nematode recovery procedures capture more than RLN. Most other nematodes, other organisms such as amoebae, fungal spores, tiny plant seeds, and organic matter are also recovered. Nematode testing labs report the number of RLN present and do the same for all other plant parasitic nematodes. All counts are to genus or family, as determining the species affiliation of every individual specimen would be prohibitively expensive.
High-quality light microscopes are used for nematode identification based on the nematode’s morphological characteristics. A clinical microscope with 40 to 100-fold magnification or a compound microscope with a 40 to 1,000-fold range is used for counting nematodes by their common name grouping. Nematodes are transparent so the shape and arrangement of internal organs as well as external physical features are used for identification.
Some labs are equipped with molecular technologies for nematode identification to the species level. Research is underway, so one day farmers may be able to order a Lesion Nematode species determination to their nematode test report.
Filtering soil mixed with water (left) collects nematodes on the bottom sieve. Incubating soil placed on top of a water reservoir (right) captures only nematodes capable of movement. Photo credit: Ann MacGuidwin


Get a Closer Look
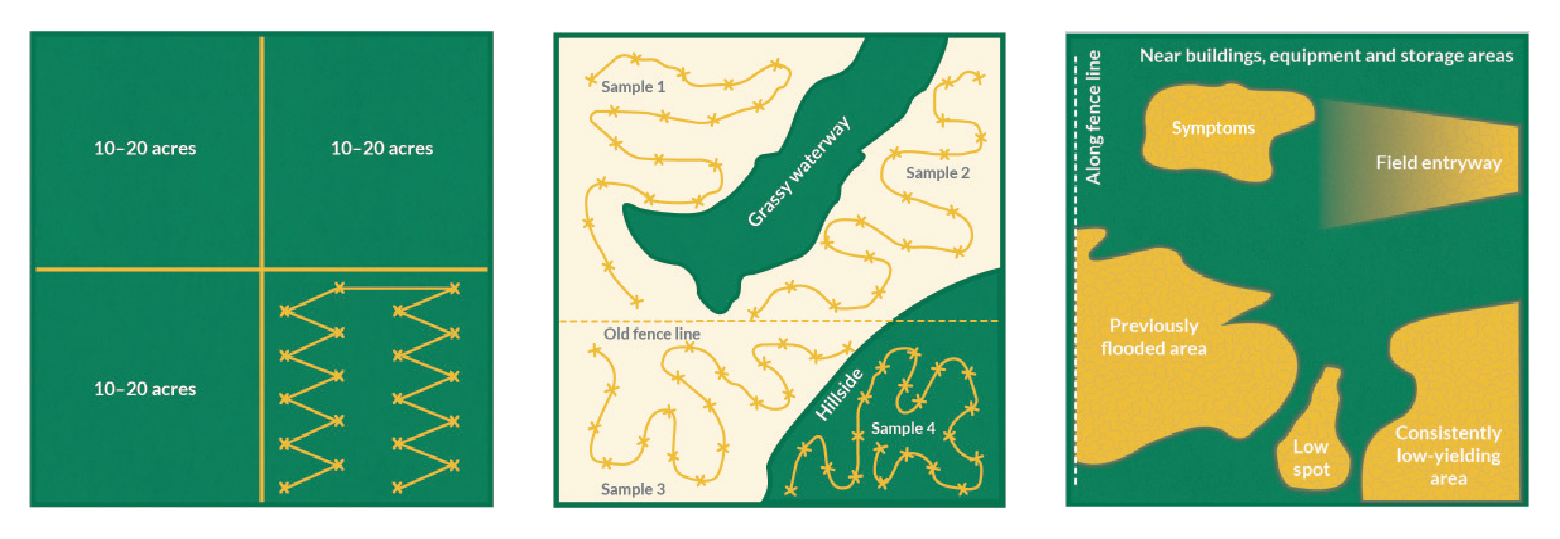
Nematode Species Detected in Soybean Fields
